
The calculator takes the elemental composition of the compound and weighs the elements to get an empirical formula mass. The molar mass of a compound is the mass in grams of 1 mole of that substance, a property expressed in units of grams per mole (g/mol)īecause the definitions of both the mole and the formula mass are based on the same reference substance, 12C, the molar mass of any compound is numerically equivalent to its formula weight in amu. Our molar mass calculator uses the periodic table and the chemical formula to solve for the molar mass of a chemical compound based on the compounds empirical formula. See original paper for the range of these elements from different sources Isotope-abundance variations and atomic weights of selected elements: 2016 (IUPAC Technical Report), Pure Appl. The masses of 1 mole of different compunds, however, are different, since the masses of the individual molecules are drastically different. List of Elements with Range of Atomic Weights. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in Pb (No3)2: Molar Mass (g/mol) Pb (Lead) 1 × 207.2 207.2. Similar to atoms, 1 mole of any compound contains the same number of molecules as 1 mole of any other compund. Do Symbol Hg Boron Carbon 5 6 15 Nitrogen 7 16 Oxygen 8 17 Fluorine 9 4.00 - Neon not round them further when performing calculations.

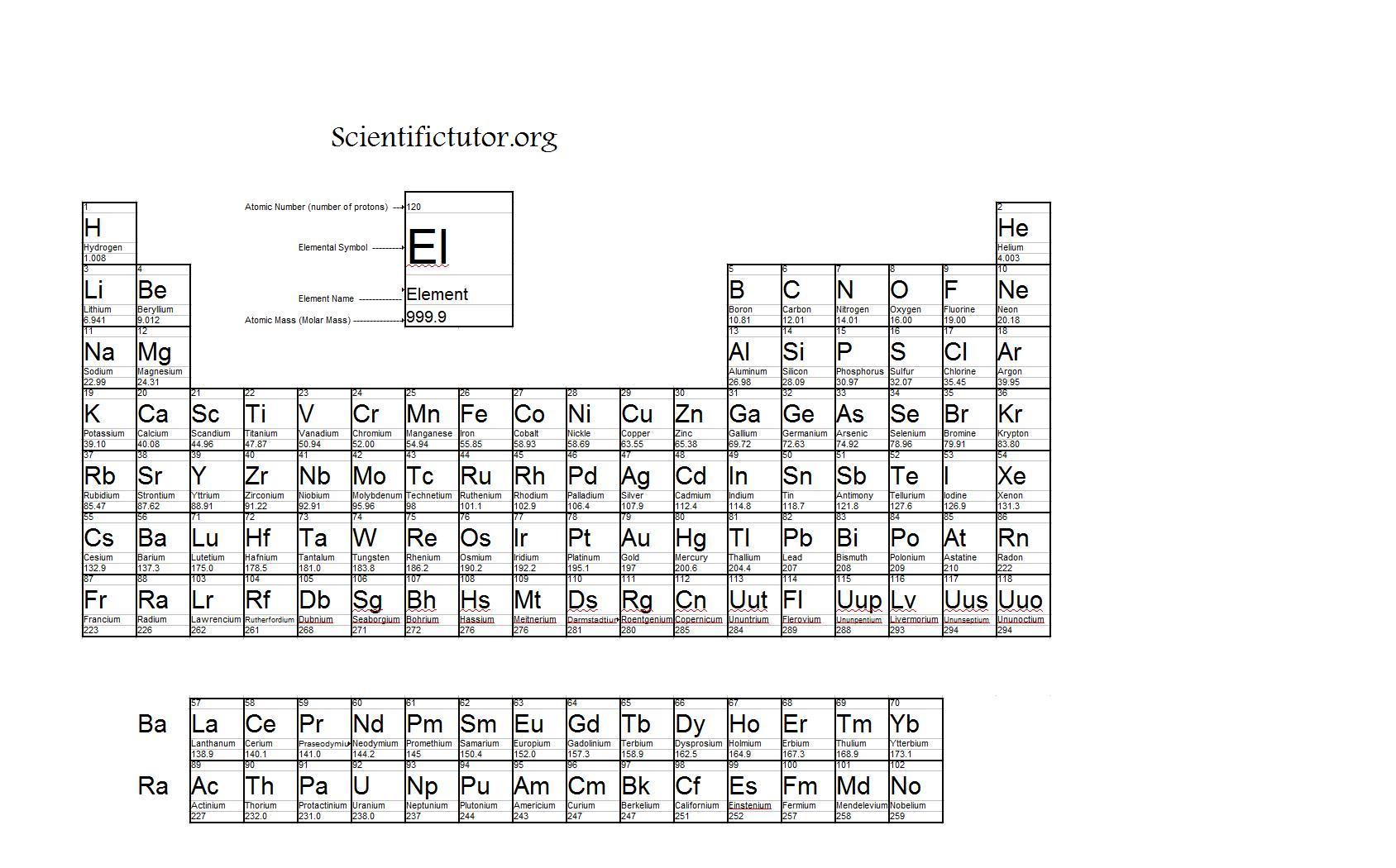
The periodic table lists the atomic mass of carbon as 12. Mercury 80 Atomic 18 Helium 2 He 13 14 All average masses are to be treated as measured quantities, and subject to significant figure rules. The conversion factors are 1 mole Cl2 over 70.90 grams Cl2, and 6.022 times 10^23 Cl2 molecules over 1 mole Cl2.\). Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in Au: Molar Mass (g/mol) Au (Gold) 1 × 196.966569 196.966569. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. It can supply you with an accurate representation of a substance’ssize and mass, and valence electron shell. Prepare a concept map and use the proper conversion factor. Periodic Table Of Elements Rounded Atomic Mass The Regular Table is a crucial part of the research into scientific research, also it can be useful in discovering a substance’s qualities. Identify the "given" information and what the problem is asking you to "find."ġ mol Cl 2 = 70.90 g Cl 2, 1mol Cl 2 = 6.022 x 10 23 Cl 2 molecules
